Abstract
Despite advances in chemotherapy and hematopoietic stem cell transplantation (HSCT), the outcome of children with relapsed acute lymphoblastic leukemia (ALL) has not significantly improved over the last 2 decades. About 50% of children with relapsed leukemia still die from their disease and ALL is still the first cause of death by cancer in children. A new hope of cure for patients with chemo-resistant cancers has emerged with the development of cancer immunotherapy. However, the major risk of post-transplant immunotherapy is the exacerbation of life-threatening Graft-versus-Host Disease (GvHD) mediated by donor-derived T cells. We therefore explored the avenue of innate immune stimulation. Several reports have demonstrated that activated Natural Killer (NK) cells can control acute myeloid leukemia (AML) in transplanted patients, whereas ALL is deemed to be resistant to NK cell killing. We recently challenged this paradigm and demonstrated that the stimulation of NK cells with third-party activated plasmacytoid dendritic cells (pDC) killed most ALL cell lines and patient-derived ALL blasts. We further demonstrated the efficacy of pDC adoptive transfers to cure ALL in a humanized mouse model of HSCT. Collectively, these results uncovered for the first time the unique therapeutic potential of activated pDC as immunotherapeutic tools to stimulate NK cell anti-leukemic activity early after HSCT. The next step toward the clinical translation of pDC-based post-transplant immunotherapy is to verify that adoptive transfers of pDC do not stimulate T cells nor exacerbate GvHD in the presence of mature T cells.
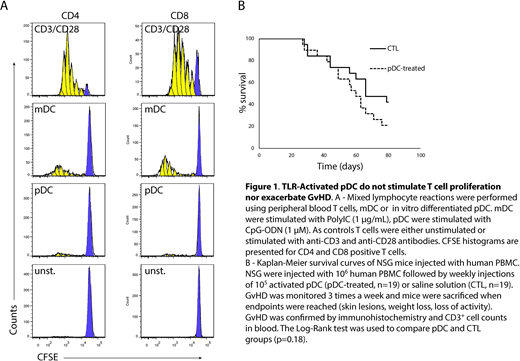
We designed a GMP-compliant method for in vitro expansion and differentiation of cord blood progenitors giving rise to sufficient numbers of pDC for adoptive transfers in patients. We showed that after Toll-like receptor (TLR) stimulation, these in vitro differentiated pDC displayed a phenotype of interferon producing cells (CD80neg PDL-1+) but not of antigen presenting cells (CD80+PDL-1neg). Accordingly, in vitro mixed lymphocyte reactions with purified allogeneic T cells demonstrated that TLR-activated pDC induced very low allogeneic T cell proliferation as compared with bona fide antigen presenting cells such as myeloid dendritic cells (mDC - CD11c+) or monocyte-derived dendritic cells (mo-DC) (Figure 1A). To test whether activated pDC could exacerbate GvHD in the presence of mature T cells, we used a xenoGvHD model in which human peripheral blood mononuclear cells (PBMC) were injected in immune-deficient mice (Nod/Scid/gRc-/-, NSG). We monitored GvHD 3-times a week according to a GvHD-assessment scale as previously described. Overt GvHD was characterized by cutaneous and intestinal lesions, weight loss and high numbers of human CD3+ cells in peripheral blood. Mice were sacrificed when endpoints were reached and GvHD was confirmed by immunohistochemistry and flow cytometry. Five weekly injections of TLR-activated in vitro differentiated pDC did not accelerate the GvHD onset and the severity of the lesions were not increased. We did not either observe any difference in survival between control and pDC-treated groups (Figure 1B).
Collectively, our results indicate that TLR-activated pDC do not stimulate allogeneic T cells and do not increase the risk of acute GvHD in a mouse model of xenoGvHD. We therefore expect this novel pDC-based immunotherapy to be safe for transplanted patients. These data open the way for the next step: a Phase I clinical trial of in vitro differentiated pDC after transplantation for leukemia.
No relevant conflicts of interest to declare.
Author notes
Asterisk with author names denotes non-ASH members.